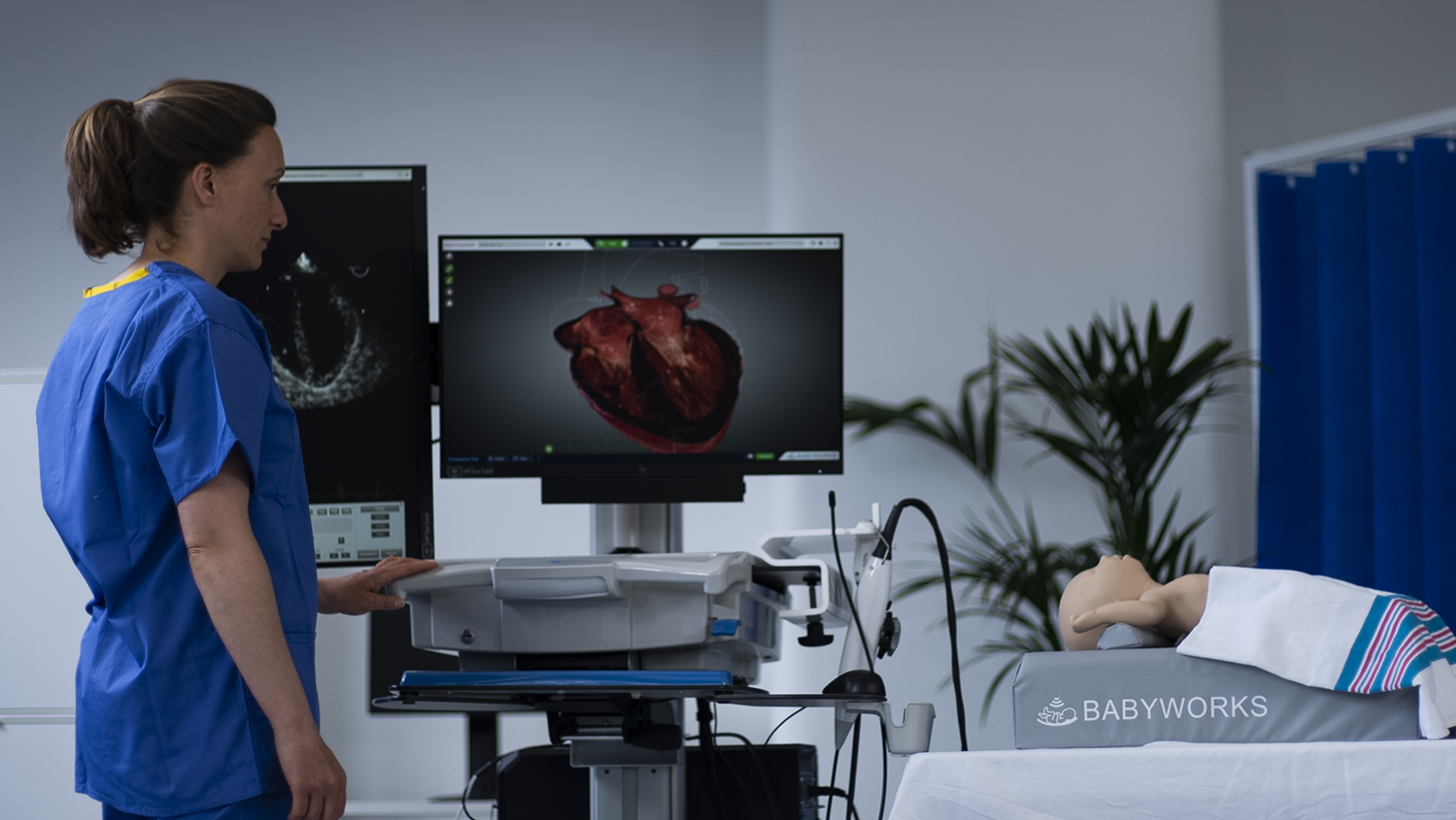
NEW updates to BabyWorks see Sam growing at pace

We’re calling for more specialist training for endometriosis scanning
Realize the potential of Point-of-Care Ultrasound (PoCUS)
Making ultrasound accessible to everyone
At Intelligent Ultrasound it’s our vision to make clinical diagnostic ultrasound easier to learn and simpler to use.
Our classroom to clinic approach reflects our belief that by supporting, guiding and speeding up scanning, we can make ultrasound more accessible to all medical professionals.
Watch our video to learn more.
From classroom…
Focussed on providing real-time ultrasound education and training through hi-fidelity simulation:

Hi-fidelity ultrasound simulators for Point-of-Care Ultrasound (PoCUS), Transesophageal and Transthoracic Echocardiography (TTE & TEE) in pediatric and neonatal care.

Hi-fidelity ultrasound simulators for Transesophageal and Transthoracic Echocardiography (TTE & TEE).

Hi-fidelity ultrasound simulators for OBGYN and general ultrasound.

Hi-fidelity ultrasound simulators for Point-of-Care Ultrasound (PoCUS) used in critical care, intensive care and emergency medicine.
…to clinic
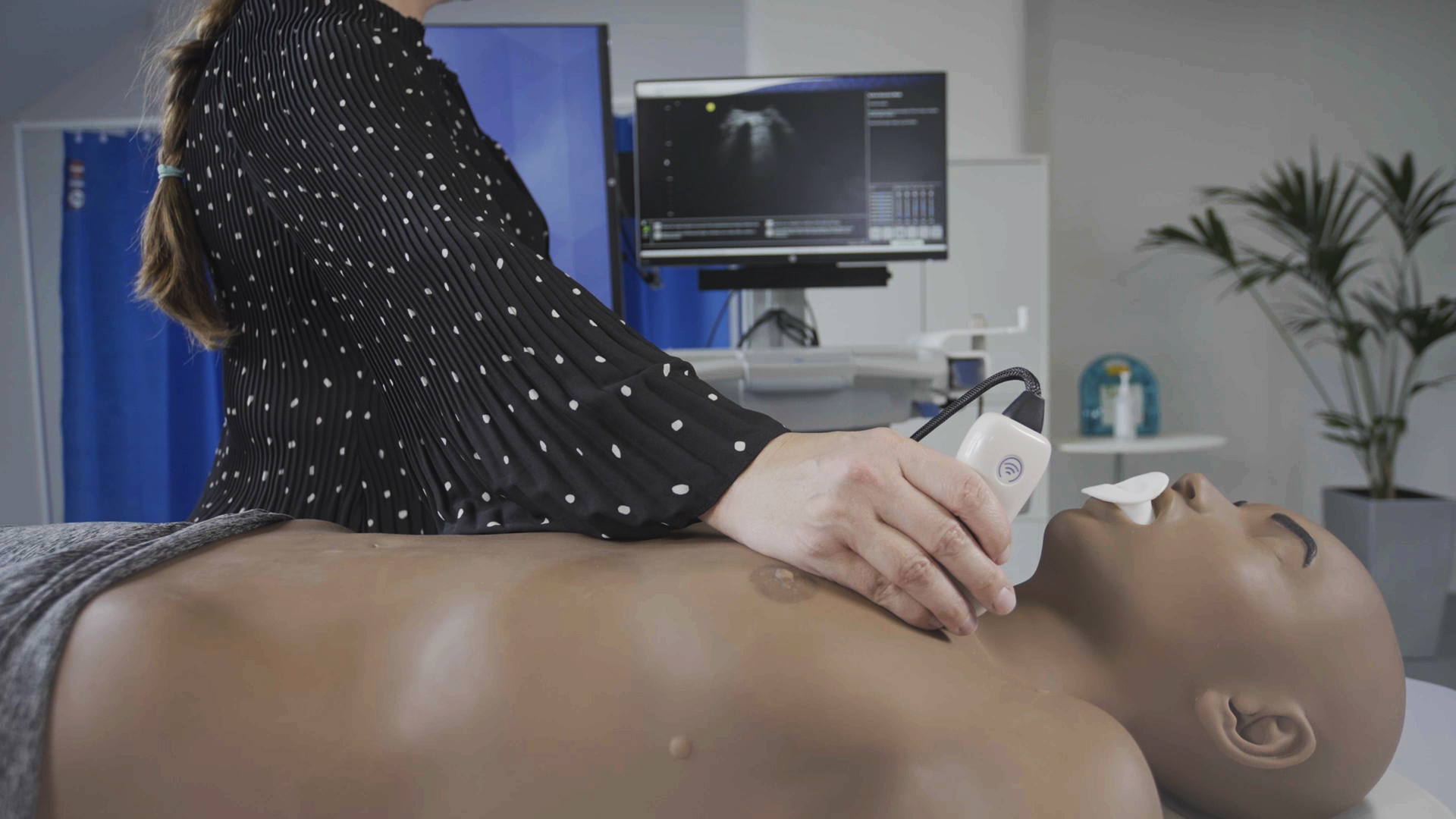
Our deep-learning-based algorithms improve real-time ultrasound image analysis and make ultrasound machines smarter and more accessible to all medical professionals.
AI-based real-time image analysis software is incorporated into GE Healthcare’s VolusonTM SWIFT ultrasound machines to support sonographers during OBGYN scans

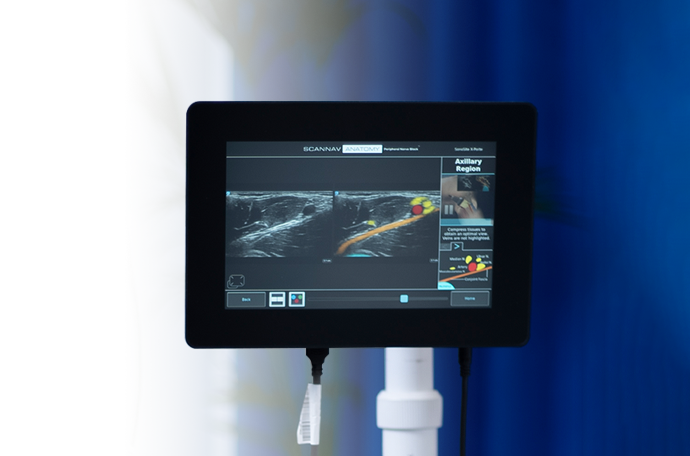
AI-based real-time highlighting of anatomy for anesthetists carrying out peripheral nerve blocks.
Since implementing ScanTrainer into our programs, not a single student has failed the transvaginal obstetrics and gynaecology module.
Naomi Brown, Senior Lecturer and Ultrasound Course Lead, University of Hertfordshire, UK
Latest News

SonoLyst, powered byIntelligent Ultrasound’s ScanNav AI, launches on new GE HealthCare Voluson Signature Series ultrasound machines
Intelligent Ultrasound announces that GE HealthCare’s SonoLyst software, powered by ScanNav AI, has been launched on the new Voluson Signature Series portfolio of ultrasound machines, the Signature 20 and Signature 18.

Revolutionizing CRNA Education with Intelligent Ultrasound Solutions
Revolutionize CRNA education with Intelligent Ultrasound Solutions, providing realistic simulation, repeatable practice, and immediate feedback. Empower CRNA students to master ultrasound procedures with confidence and competence for real-world practice.

The Synergy of BodyWorks Eve and HeartWorks
While both BodyWorks Eve and HeartWorks excel in their respective domains, it's their synergy that truly sets them apart, allowing learners to transition between different areas of learning within one unified platform that maximizes efficiency and effectiveness.